Summary
Colostrum is nature's perfect first food. It is the pre-milk substance that emerges from the mother's breast following the birth of the newborn. Colostrum is a mixture of immune and growth factors as well as important nutrients, all designed to activate newborn's immune system, insuring the health, vitality, and growth of the newborn. Numerous studies show that colostrum and its components continue to exert important biological activities also when given to adults, so that its beneficial effects extend well beyond the neonatal period of development. Over the last sixty years or so, scientists have begun to untangle the complicated web of colostrum, teasing out the many different components that make it up. It has become clear that many of the substances in colostrum function as signal transducers that turn on or off different cell functions, particularly those of the immune system. These signaling molecules are called proline-rich polypeptides (PRPs) and Apelin peptides (APs) and are the most important components in colostrum. Modulating cytokine production from the activated T-cells and macrophages, PRPs have the unique ability to balance the immune system, turning it up when needed to fight an infection, or turning it down when it is overactive, such as is the case in autoimmune disorders.
Introduction
Colostrum is the first complete food available to newborn mammals and is essential for their life. It provides many important biological nutrients passed on from the mother's blood. Colostrum is defined as the pre-milk that is in the breast or udder when the baby or calf is born. Milk produced immediately after birth to day five after birth is called "Transition Milk." After day five, milk is defined.
Colostrum contains various immune factors, such as immunoglobulins (Ig), proline-rich polypeptides (PRPs) and lactoferrin (LF), as well as important growth factors for developing healthy muscles and bones. These components are illustrated in Figure 1. Colostrum activates and regulates numerous immune, growth, and repair processes.
colostrum-keech-figure1.jpg
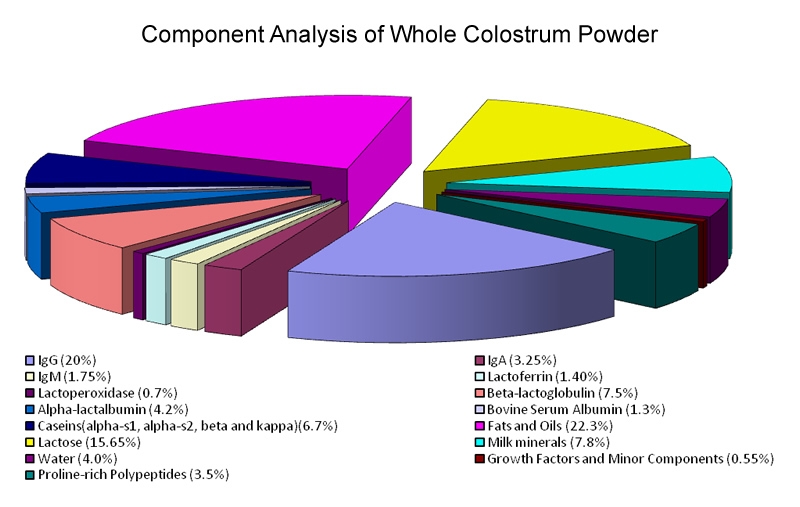
Image provided by Dr. Keech.
Figure 1: Pie Chart showing components in dried form of first milking bovine colostrum
The protein content of bovine colostrum is three to four times higher (up to 150 g/L compared to 30 to 40 g/L found in regular cow's milk. The greater part of this protein content is comprised of the immunoglobulins whey proteins.
|
Time after Birth (hr) |
Total (i) Protein % |
Casein % |
Albumin % |
Fat % |
Lactose % (ii) |
Ash % |
Total Solids % (iii) |
|---|---|---|---|---|---|---|---|
|
0 |
17.57 |
5.08 |
11.34 |
5.10 |
2.19 |
1.01 |
26.99 |
|
6 |
10.00 |
3.51 |
6.30 |
6.85 |
2.71 |
0.91 |
20.46 |
|
12 |
6.05 |
3.00 |
2.96 |
3.80 |
3.71 |
0.89 |
14.53 |
|
24 |
4.52 |
2.76 |
1.48 |
3.40 |
2.98 |
0.86 |
12.77 |
|
36 |
3.98 |
2.77 |
1.03 |
3.55 |
3.97 |
0.83 |
12.22 |
|
72 |
3.86 |
2.70 |
0.97 |
3.10 |
4.37 |
0.84 |
11.86 |
Table 1: Chemistry of raw liquid bovine colostrum from 0-72 hours after parturition
Colostrum contains cytokines and other protein compounds of very low molecular weight that act as biologic response modulators (BMR's), which intervene locally in most biological processes. (ii), Lactose has a great influence on allergic-like responses experienced by some individuals due to the lack of lactase and B-galactosidase. Unlike immunoglobulin IgG, lactose rises in value from the first milking through the subsequent several milkings; (iii), Total solids are determined by the dried solids from the original quantity of liquid started with.
Source: Immuno Dynamics, Inc. ICN Biomedical, Inc. Cosa Mesa, CA.
The major components of colostrum
The most important components of colostrum can be broken down into three major categories: Immune System Factors, Growth Factors, and Nutritional Components.
Immune System Factors
Immunoglobulins
Immunoglobulins are glycoproteins and are present in serum and other tissue fluids, including the milk and colostrum, of all mammals and have an antibody function.
Antibodies are produced in response to the host being exposed to immunogenic foreign substances (antigens), such as infectious microbes.
Bovine immunoglobulins are a group of bioactive molecules, all of which work together to fight disease and destroy invading pathogenic microbes. The immunoglobulin fraction in bovine colostrum is composed of approximately 70-85% IgG, 10-15% IgA, 10-15% IgM, while IgE and IgD are found in small amounts.
Immunoglobulin G (IgG) — The predominant antibody found in bovine colostrum, making up about 80-85% of the total immunoglobulins. Human colostrum contains significantly less IgG and much more IgA than does cow colostrum due to the differences in human and cow placentas.
IgG imparts systemic immunity. That is, it can impart immunity in one individual if taken from another individual who already has immunity to a certain pathogen. This is how a mother cow imparts her own immunity to her calf and it is also how local intestinal immunity can be transferred from a cow to a human via colostrum. After taking bovine colostrum, IgG stays within the human digestive tract, and does not enter the human's blood stream.
Immunoglobulin A (IgA) — The predominant immunoglobulin in human milk and colostrum. The primary role of IgA is as first line of defense, protecting mucosal surfaces and preventing the attachment of pathogens to them. This immunoglobulin is mainly involved with local immunity against pathogens. The type of IgA in colostrum is called secretory IgA (S-IgA) because it has a protective protein attached to it to protect against digestive enzymes, to help the molecule cross the intestinal lining, and to prevent the attachment of pathogens to mucosal surfaces in the gastrointestinal tract.
Its main function in colostrum appears to be to prevent gastrointestinal infections. For example, an immunoglobulin preparation from bovine colostrum that contains IgA reacts against toxins associated with E. coli and Shigella infections in cell cultures in vitro (1).S-IgA from human colostrum also inhibits adherence of Vibrio cholera to intestinal tissue in vitro, an activity that is consistent with the ability of IgA to prevent bacterial colonization.1 S-IgA and IgM autoantibodies from human colostrum also protect in vitro against GI autoantigens that can cause autoimmune disease, and thus act to maintain immune homeostasis.2,3 Results from three separate clinical studies also show that oral immunoglobulins from bovine colostrum are anti-diarrheal in adult AIDS/HIV patients suffering from infection by Cryptosporidium parvum (5-7). In other biological activities, orally administered S-IgA enriched anti-measles bovine colostrum has shown a weak effect in multiple sclerosis patients.4 A follow-up double blind study, however, found that hyperimmune bovine colostrum provided no symptomatic relief to such patients.5 S-IgA also inhibits cytokine release in vitro, indicating that it may play an anti-inflammatory role.6
Immunoglobulin M (IgM) — IgM enhances phagocytosis by destroying invading pathogens. Basically five IgG like monomer structures strung together in a circular arrangement, IgM is the first antibody produced in response to an antigen and its primary role is "first to fight."
Immunoglobulin E (IgE) — IgE is involved in the allergic reaction and histamine-associated allergic reactions, as well as active defense against enteric parasites. Involved in allergic responses to antigens. It binds to mast cells and basophils, specialized cells in the body that participate in allergic reactions. When an antigen comes into contact with IgE, the mast cell or basophil releases histamine and other chemicals which produce the allergic reaction.7 IgE is also highly antiviral.
Immunoglobulin D (IgD) — IgD attached to B cells; acts as an antigen receptor on B cells. It stimulates lymphocytes to produce antibodies by presenting antigens to them. It is also highly antiviral.8
Proline-rich Polypeptides (PRPs) and Apelin Peptides (APs)
PRPs (also often called info-peptides, info-proteins, or cytokine precursors) exist naturally in the human body only in small amounts and are the most important immune modulating component in colostrum. Peptides are short chains of amino acids (500-2900MW) without the complex tertiary structure of proteins. Proline-rich polypeptides, so-called because they contain an unusually high proportion of the amino acid proline, are intercellular signaling molecules that have the unique ability to modulate the immune system, turning it up when needed to fight an infection, or turning it down when it is overactive, such as is the case in autoimmune disorders.
These naturally occurring peptides are found throughout the body and are the main regulators of cell protein synthesis.
Apelin Peptides are a larger class of colostrum peptides (1400-7000MW)9. Some of these APs are neuropeptides involved in the regulation of body fluid homeostasis and cardiovascular functions. APs reduces blood pressure via a nitric oxide-dependent, possibly central mechanism, exerting a stronger positive inotropic effect. Apelin is widely distributed in the Central Nervous System and periphery, especially in the heart, kidney, lung and mammary glands. Apelin-like immunoreactivity is detected in the adipocytes, gastric mucosa, endothelia and Kupffer cells in the liver. Apelin Receptors are G protein-coupled receptors (GPCRs) which bind apelin peptides on the T-cells. APs have shown to block the entry of the human Immunodeficiency virus (HIV)10
Cytokine Production
Cytokines are produced by the activated macrophages and activated T helper cells which act as cytokine factories directing the correct information to other immune cells in the process of an immune response (Fig 2). The most important action of the PRPs is to modulate the production of all the cytokines; and they do this by binding to specific PRP receptor sites on these activated T helper cells and activated macrophages (the center cells in Figure 2).
colostrum-keech-figure2.jpg
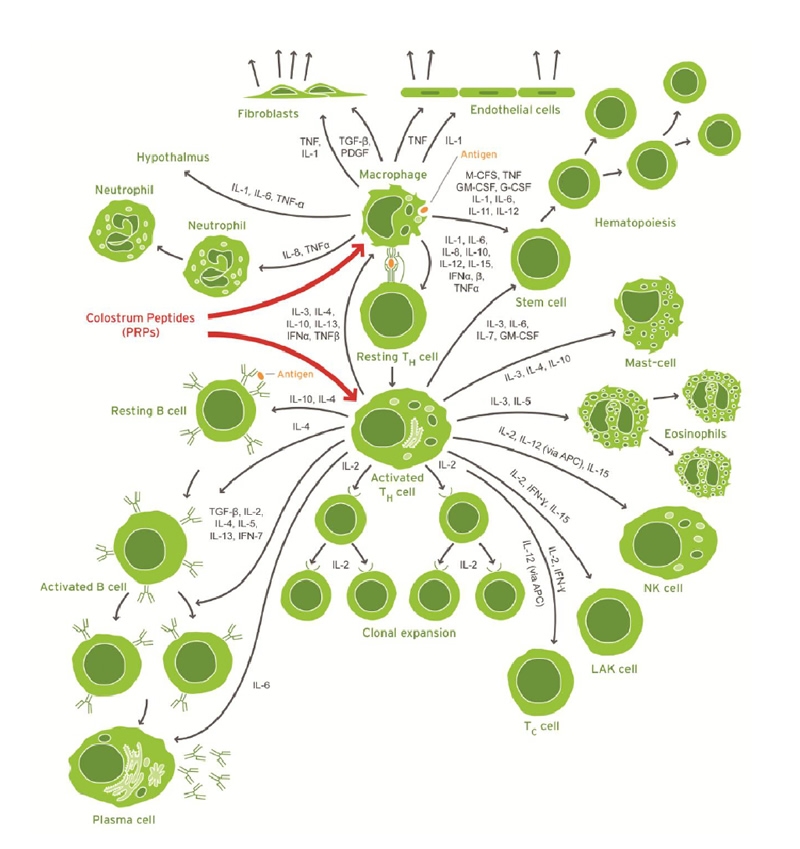
Image provided by Dr. Keech.
Figure 2 : Directed cytokine response by human activated macrophages and T helper cells
The activated macrophages and T helper cells direct the correct information to other immune cells in the process of an immune response. As cytokine precursors, the PRPs specifically attach to receptor sites on the activated T helper cells and activated macrophages to modulate the cytokine production.
The activated macrophages and T helper cells direct the correct information to other immune cells in the process of an immune response. As cytokine precursors, the PRPs specifically attach to receptor sites on the activated T helper cells and activated macrophages to modulate the cytokine production.
In homeostasis, the levels of all cytokines are balanced. If the immune system is under attack by pathogens, allergens, viruses, bacteria, fungi, toxins, etc., the body produces a cytokine cocktail in an attempt to direct an appropriate immune response. Some invaders trick the immune system to a state of constant over activity, as in the case, for example, of a "cytokine storm" for H1N1 (swine flu) infections.
All diseases in the human body are initially the result of a cytokine imbalance manifested in the form of symptoms associated with that disease/condition. An overproduction of autoimmune specific cytokines is classified as T-lymphocyte Type I (Th1) state and an overproduction of allergy cytokines is classified as T-lymphocyte Type II (Th2) state (Fig 3).
An immunologist solution to reversing these disease states is to rebalance the TH2/TH1 cytokine production by the activated macrophages and T helper cells.
Numerous studies have shown PRP supplementation from bovine colostrum can help reset and rebalance the cytokine production.
colostrum-keech-figure3.jpg
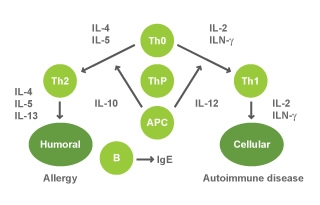
Image provided by Dr. Keech.
Figure 3 : Cytokines influencing T-cell maturation in an allergic or autoimmune response
Balancing the immune system with PRPs from colostrum is not only important in newborns just beginning to live in a hostile environment, but also in adults with over- or underactive immune systems, perhaps resulting from various types of stress. Like many drugs, PRPs attach to the G-protein Coupled Receptors (GPCR) on the surfaces of T-cells. As cytokine precursors, PRPs can help stimulate an underactive immune system or down regulate an overactive immune system as seen in autoimmune disease (MS, rheumatoid arthritis, lupus, scleroderma, chronic fatigue syndrome, allergies, etc.). They have been shown to improve or eliminate symptoms of both allergies and autoimmune conditions, such as pain, swelling, and inflammation. PRPs act both in vitro and in vivo, and are not species specific.
PRPs have also been tested clinically in the treatment of Alzheimer's disease (AD). Orally administered PRP complex from sheep colostrum (100 mg every other day) was found to significantly improve Alzheimer's dementia in a double blind, placebo-controlled study11. All 15 AD patients stabilized or improved; none of the 31 selenium-treated or placebo control patients improved.
Cytokines
Cytokines can be classified as proteins, peptides or glycoproteins that exert either a pro- or anti-inflammatory effect on the immune system. Colostrum is known to stimulate the production of cytokines in peripheral blood cells.12 Cytokines found in colostrum include interleukin-1 (IL-1),13 IL-1b,14 IL-1ra,15 IL-3, IL-4,16 IL-5,17 IL-6,18 IL-8,19 IL-10,20 IL-12,21 IL-13, IL-16, IL-18,22 interferon-gamma (INF-γ),23 tumor necrosis factor24 alpha (TNF-α),25 and TNF-γ receptors.
Cytokines are any of several regulatory proteins, such as the interleukins and lymphokines, that are released by cells of the immune system and act as intercellular mediators in the generation of an immune response. Cytokines are produced in the human body by the actions of PRPs. Two main cytokines that are produced by PRPs are IFN-α and IFN-β. Cytokines IL-1, -6, interferon-γ, and lymphokines stimulate the lymph glands and are thought to be highly effective antiviral immune substances. Interleukins that regulate the duration and intensity of the immune response are responsible for cell-to-cell communication and boost T cell activity and the production of immunoglobulins. Interleukin-10 is strongly anti-inflammatory, especially in arthritic joints. The benefits of cytokines in the treatment of cancer was first popularized by the 1985 Steven Rosenberg Book, Quiet Strides in the War on Cancer. Since that time, the same cytokines found in colostrum (IL-1, 6, 10, interferon-γ, leukocytes, lymphokines, tumor necrosis factor) have been the most researched protocols in scientific research for the cure for cancer. The term cytokine, or immunocytokines, was used initially to separate a group of immunomodulatory proteins, also called immunotransmitters, from other growth factors that modulate the proliferation and bioactivities of non-immune cells. However, this terminology suggesting a clear-cut distinction cannot be maintained and may not be altogether meaningful. Some cytokines are produced by a rather limited number of different cell types while others are produced by almost the entire spectrum of known cell types. Today the term cytokine is used as a generic name for a diverse group of soluble proteins and peptides that act as humoral regulators at nano to picomolar concentrations and that, either under normal or pathological conditions, modulate the functional activities of individual cells and tissues. These proteins also mediate interactions between cells directly and regulate processes taking place in the extracellular environment.
Other immune factors
Lactoferrin (LF) — LF has potent anti-bacterial, anti-viral, and anti-fungal activities to help protect the body from invading pathogens. It is also secreted in milk, tears, mucus, and saliva, particularly in response to a pathogen attack. Many of the biological activities of LF relate to its ability to regulate either the immune system26,27 or the availability of iron. LF is an iron-binding protein that helps deprive bacteria of the iron they require to reproduce and releases iron into the red blood cells enhancing oxygenation of tissues.
Lysozyme — An enzyme that is capable of degrading the outer membrane of gram-positive bacteria. It also acts in concert with lactoferrin to kill gram-negative bacteria. Lysozyme is also a hydrolyzing agent and immune system booster capable of destroying bacteria and viruses on contact.
Lactoperoxidase — Another enzyme which, in the presence of hydrogen peroxide (formed naturally in the body), catalyzes the oxidation of thiocyanate, a component of saliva, to form hypothiocyanate, which can kill both gram-positive28 and gram-negative29 bacteria.
Lactalbumin — Research indicates that lactalbumins may be highly effective against numerous forms of cancer and viruses. Colostral lactalbumin has been found to be able to cause the selective death (apoptosis) of cancer cells, leaving the surrounding noncancerous tissues unaffected.
Lactobacillus Bifidus & Acidophilus — Friendly flora, which are necessary for the digestion of food and in the reduction of the growth of harmful bacterial in the digestive system. They have been shown to effectively combat Candida albicans.
Enzymes — Lactoperoxidase-thiocyanate, peroxidase and xanthine oxidase oxidize bacteria through their ability to release hydrogen peroxide.
Digestive Enzymes and Proteins — Colostrum contains proteins that inhibit the action of digestive enzymes, allowing the contents of colostrum to reach their targets without significant degradation.30 Protease inhibitors also facilitate the passage of macromolecules through the gut lining, which is important in the newborn.31 Colostrum also contains various enzymes that help the components of colostrum do their job. Enzymes basically function to facilitate chemical reactions in the body. Trypsin Inhibitors and Protease Inhibitors prevent the destruction of immune and growth factors in colostrum from being broken down in the GI tract. They also prevent H. pylori from attaching to the walls of the stomach and can have a beneficial role in the treatment of peptic ulcers.
Neurotransmitters — Endogenous signaling molecules such as leptin that alter the behavior of neurons or effector cells.
Soluble CD14 — CD14 or endotoxin receptor is found in a soluble form in colostrum and milk where it acts to directly induce B cell growth and differentiation, thus enhancing IgG secretion.32
Cathelicidin-derived Antimicrobial Peptide — Certain peptides of the cathelicidin family of peptides found in milk and colostrum have antimicrobial activity against both gram-positive and gram-negative bacteria. 33
Nucleotides — Dietary nucleotides, the building blocks of DNA and RNA, enhance dietary growth and maturation as well as humoral and cellular immunity.34 Infants who receive human milk with nucleotides have higher antibody levels, lower rates of diarrhea, and higher natural killer cell activity.35,36
Cardiovascular Health Components — A number of components of colostrum are important to cardiovascular health, including ACE inhibitors that help regulate blood pressure, and clotting factors.
Growth Factors
Growth factors enhance wound healing, cartilage formation, and bone repair, stimulate muscle protein synthesis/inhibit protein breakdown, benefit GI tract structure and function, and act on the immune system to inhibit tissue inflammation and allergic response.
Colostrum, as might be expected, contains many growth factors. Their presence in colostrum is primarily to complete the growth and development of the newborn gut, but in older children and adults they act to repair damage to the gut lining that might result in leaky gut syndrome as well as other health maintenance functions.37,38 Their main function in the organism is to regulate the growth and development of cells.39 They play a major role in wound healing and the repair of broken bones40 as well as injured tendons and ligaments.41 Growth factors, just like PRPs, are generally peptides that function as intercellular signaling molecules to turn on or turn off the production of specific proteins in the target cells. The body's growth factors are capable of increasing T cell production, accelerating healing, balancing blood glucose, reducing insulin need, increasing growth, and repair of vital tissues while metabolizing fat for fuel. Medical studies have shown the vital growth factors IgF-1, TGF-α and TGF-β, and nucleotides from bovine colostrum to be identical to human in composition. They help provide the raw materials to repair vital DNA and RNA in the body's cells. By stimulating DNA formation, it has been shown that they can help stimulate normal cell and tissue growth, regeneration, and accelerated repair of aged or injured muscle, skin collagen, bone, cartilage, nerve tissues, heart muscle and new blood vessels for collateral coronary circulation. These growth factors facilitate the healing of tissues damaged by ulcers, trauma, burns, surgery, or inflammatory disease.
Insulin-like Growth Factor 1 (IGF-1) — Originally known as somatomedin C. It is very similar in structure to insulin. IGF-1 affects nearly every cell in the body, in particular muscle cells, cartilage, bone, liver, kidney, nerves, skin and lungs.42 It regulates cell growth and development as well as cellular DNA synthesis. Studies have shown that both TGF-b and IGF-1 speed the healing of bones, which is particularly important in the elderly whose bones heal very slowly.43 IGF-1 also is responsible for increasing muscle mass in response to muscle over loading.44 IGF-1 has also shown promise in peripheral nerve regeneration.45 In the newborn, IGF-1 plays a critical role in the development of the newborn gut.46
Human trials in 1990 reported that IGF-1 stimulates glucose utilization. It can help balance blood sugars (non-insulin diabetics and hypoglycemia). Inadequate levels of IGF-1 are associated with an increased incidence of Type 2 diabetes and difficulty in losing weight despite a proper nutritional intake and adequate exercise.
Insulin-like Growth Factor (IGF-2) — Very similar in function to IGF-1. Its major function is to promote growth during fetal development.47
Both IGF-1 and IGF-2 promote the growth of arterial cells, and disruption of the dynamic balance between IGF-1, IGF-2 and IGF binding proteins has been implicated in the formation of atherosclerotic plaques in the arteries and resulting restenosis (narrowing of the artery).48
Transforming Growth Factor-alpha (TGF-α) — Induces epithelial development49,50 and stimulates neural development in the injured brain.51 It is closely related to EGF.
Transforming Growth Factor-beta (TGF-β) — Only TGF-β1 and β2 are found in human and bovine milk and colostrum. TGF-β controls cellular proliferation and differentiation.52 It stimulates the production of IgA by B lymphocytes.53,54 It is particularly important in the development of cartilage and bone.55,56
Epidermal Growth Factor (EGF) — Stimulates the proliferation and differentiation of epidermal cells, including the lining cells of the gut that maintain the gut integrity and the proper permeability.57,58 It exerts its effects by binding to the EGF receptor protein in the cell membrane of the target cell.59 It has been used to promote wound healing by stimulating cell proliferation in the wound.60
Fibroblast Growth Factor (FGF) — Fibroblasts are the cells in the connective tissue that help to promote wound healing by stimulating cell proliferation in the wound. It also helps maintain normal bone and repair fractures.61,62 It also plays a role in the healing of ulcers.
Nutritional Components
Vitamins and Minerals — Colostrum is not a supplement, it is a whole food for the newborn. It is a combination of vitamins and minerals which are naturally occurring in a perfect combination. Vitamins A, B12, and E are found in small amounts, while traces of all others are also present in colostrum.
Oligopolysaccharides and Glycoconjugates — Attract and bind to pathogens, such as Streptococcus, E. coli, Salmonella, Cryptosporidia, Giardia, Entamoeba, Shigella, Clostridium difficile (toxins A & B), preventing them from attaching or entering the mucous membranes.
Glycoproteins — Colostrum contains a number of complex carbohydrates, including glycoproteins, glycolipids, glycosaminoglycans, mucins, and oligosaccharides, which are basically complex sugars either alone or in combination with proteins or lipids. They are known to inhibit pathogen binding to gut lining cells, preventing their entry into the bloodstream, usually by competing for binding sites on the cell membranes. One fucosyl oligosaccharide inhibits E. coli toxin. Another inhibits Campylobacter jejuni, which is a major cause of gastroenteritis and food poisoning. Oligosaccharides inhibit the binding of Streptococcus pneumoniae, which causes many infections such as pneumonia, meningitis, otitis, osteomyelitis, peritonitis and endocarditis, and enteropathogenic E. coli, which causes diarrhea. A mannosylated (mannose is a type of sugar) glycopeptide inhibits the binding of enterohemorrhagic E. coli, which causes bloody diarrhea. One of the glycosaminoglycans prevents the binding of HIV structural protein to CD4+cells (T cells), which is the first step in HIV infection. Mucin, found in milk and colostrum, inhibits the binding of S-fimbriated E. coli (fimbriae are thread-like structures used by certain bacteria to adhere to cells). Ganglioside GM1, a membrane protein, reduces diarrhea due to cholera toxin and E. coli labile toxin. Glycosphingolipid Gb3, another membrane protein, binds to shiga toxin, a toxin produced by Shigella dysenteriae (cause of dysentery) and the shigatoxigenic group of E. coli. Shiga toxin functions much like the deadly toxin ricin by inhibiting protein synthesis in target cells.63
Kappa-caseino glycomacropeptide — This is a peptide from the casein fraction of milk and colostrum that interferes with the binding of viruses and bacteria in the intestine, binds cholera and E. coli toxins, promotes the growth of beneficial bacteria in the gut, and helps modulate the immune system (66-68).64,65,66
Fatty acid Binding Protein — A member of the lipocalin family of secreted proteins, fatty acid binding protein regulates two molecular pathways in macrophages that coordinate inflammatory activity and cholesterol uptake.67
Beta-lactoglobulin — Another member of the lipocalin family of proteins, β-lactoglobulin has antimicrobial and antiviral activity.68,68
Orosomucoids — Increased levels of this glycoprotein are associated with inflammation. It acts as an anti-inflammatory mediator, particularly in the endothelium of capillaries where it inhibits the effect of histamine.70,71
Clusterin (Apolipoprotein J) — Clusterin plays an important role in cell-cell and cell-substratum interactions.72 It also acts as an extracellular molecular "chaperone" that "steers" proteins into cells.73
Fats — Although normally defatted when converted into a powder, whole colostrum also contains fats, including phospholipids, fatty acids (linoleic acid, dihomo-gamma- linoleic acid, alpha-linoleic acid, octadecatetraenoic acid, eicosatrienoic acid, docosahexaenoic acid, docosapentaenoic acid, and arachidonic acid), saposins (A, B, C, D), prosaposin, tocopherols, and cholesterol (76-78).74,75,76. These are necessary nutrients for the newborn, while in the adult, milk fat in whole milk is believed to be beneficial to the regulation of blood sugar levels and appetite.77
Colostrum and its components exert their biological activities when given orally. Communication with the immune system via the intestinal mucosa is believed to be important in initiating many of colostrum's immunomodulatory activities. Moreover, the major composition of bovine (cow) and human colostrum is similar, and colostrum from both sources is biologically active.
Conclusions
In conclusion, the most important component in colostrum that acts to regulate and balance the immune system are the PRPs. Studies suggest that this biological activity is important for adults with over- or under-active immune systems as well as for newborns. Colostrum and its components also have significant anti-bacterial, anti-viral, and antifungal activities. Colostrum and its growth factors enhance muscle protein synthesis, wound healing, cartilage formation, and bone repair, benefit GI tract structure and function, and are anti-inflammatory and anti-allergenic. Colostrum and its components exert their biological activities when given orally, in many cases as a dietary supplement. Moreover, bovine and human colostrum are similar enough that they exert similar biological activities in various species.
(A copy of Dr Keech's book Peptide Immunotherapy, Colostrum: A Physician's Reference Guide, can be obtained by visiting Dr. Keech's website.)
References
1 Majumdar AS, Ghose AC (1982) Protective properties of anticholera antibodies in human colostrum. Infect Immun 36(3) 962-965.
2 Vassilev TL, Veleva KV (1996) Natural polyreactive IgA and IgM autoantibodies in human colostrum. Scand J Immunol 44(5) 535-539.
3 Quan CP, Berneman A, Pires R et al (1997) Natural polyreactive secretory immunoglobulin A autoantibodies as a possible barrier to infection in humans. Infect Immun 65(10) 3997-4004.
4 Ebina T, Sato A, Umezu K et al (1984) Treatment of multiple sclerosis with anti-measles cow colostrum. Med Microbiol Immunol (Berl) 173(2) 87-93.
5 Bayard BL, James MA (1987) Hyperimmune bovine colostrum inefficacious as multiple sclerosis therapy in double-blind study. J Am Diet Assoc 87(10) 1388-1390.
6 Larson RE, Ward AC et al (1974) Capability of lambs to absorb immunoproteins from freeze-dried bovine colostrum. Am J Vet Res 35(8) 1061-1063.
7 Thatcher EF, Gershwin LJ (1989) Colostral transfer of bovine immunoglobulin E and dynamics of serum IgE in calves. Vet Immunol Immunopathol 20(4) 325-334.
8 Keller MA, Heiner DC, Myers AS, Reisinger DM (1985) IgD in human colostrum. Pediat Res 19(1) 122-126.
9 Mesmin et al. 2011, J Proteome Res 10 (11): 5222-5231.
10 Zou et al. FEBS Lett 2000 May 4:473(1): 15-8.
11 Leszek J, Inglot AD, Janusz M, Lisowski J, Krukowska K Georgiades A J (1999) Colostrinin: a proline-rich polypeptide (PRP) complex isolated from ovine colostrum for treatment of Alzheimer's disease. A double-blind, placebo-controlled study. Arch Immunolog Therap Experiment 47 377-385.
12 Bessler H, Straussberg R, Hart J, Notti I, Sirota L (1996) Human colostrum stimulates cytokine production. Biol Neonate 69(6) 376-382.
13 Bocci V, von Bremen K, Corradeschi F, Luzzi E, Paulesu L (1991) What is the role of cytokines in human colostrum? J Biol Regulators Homeostatic Agents 5(4) 121-124.
14 Goto M, Maruyama M, Kitadate K, Kirisawa R, Obata Y et al (1997) Detection of interleukin-1 beta in sera and colostrum of dairy cattle and in sera of neonates. J Vet Med Sci 59(6) 437-441.
15 Hagiwara K, Kataoka S, Yamanaka H, Kirisawa R, Iwai H (2000) Detection of cytokines in bovine colostrum. Vet Immunol Immunopathol 76(3-4) 183-190.
16 Zizka J, Kverka M, Novotna O, Stankova I, Lodinova-Zadnikova R et al (2007) Perinatal period cytokines related to increased risk of future allergy development. Folia Microbiol (Praha) 52(5) 549-555.
17 Zizka, 2007, ibid.
18 Rudloff HE, Schmalstieg FC Jr, Palkowetz KH, Paszkiewicz EJ, Goldman AS (1993) Interleukin-6 in human milk. J Reproduct Immunol 23(1) 13-20.
19 Hashira S, Okitsu-Negishi S, Yoshino K (2002) Interleukin 8 in the human colostrum. Biol Neonate 82(1) 34-38.
20 Garofalo R, Chheda S, Mei F, Palkowetz KH, Rudloff HE et al (1995) Interleukin-10 in human milk. Pediatr Res 37(4pt1) 444-449.
21 Bryan DL, Hawkes JS, Gibson RA (1999) Interleukin-12 in human milk. Pediatr Res 45(6) 858-859.
22 Takahata Y, Takada H, Nomura A, Ohshima K, Nakayama H et al (2001) Interleukin-18 in human milk. Pediatr Res 50(2) 268-272.
23 Bocci (1991) op. cit.
24 Buescher ES, McWilliams-Koeppen P (1998) Soluble tumor necrosis factor-alpha (TNFalpha) receptors in human colostrum and milk bind to TNF-alpha and neutralize TNF-alpha bioactivity. Pediatr Res 44(1) 37-42.
25 Bocci, 1991, op. cit.
26 Wlasczyk A, Zimecki M, Adamik B et al (1997) Immunological status of patients subjected to cardiac surgery: effect of lactoferrin on proliferation and production of interleukin 6 and tumor necrosis factor alpha by peripheral blood mononuclear cells in vitro. Arch Immunol Ther Exp (Warsz) 45(2-3) 201-212.
27 Zimecki M, Wlaszczyk A, Cheneau P et al (1998) Immunoregulatory effects of a nutritional preparation containing bovine lactoferrin taken orally by healthy individuals. Arch Immunol Ther Exp (Warsz) 46(4) 231-240.
28 Steele WF, Morrison M (1969) Anti-streptococcal activity of lactoperoxidase. J Bacteriol 97(2) 635-639.
29 Björck L, Rosen C, Marshall V, Reiter B (1975) Antibacterial activity of the lactoperoxidase system in milk against pseudomonads and other gram-negative bacteria. Appl Microbiol 30(2) 199-204.
30 Quigley, JD III, Martin KR, Dowlen HH (1995) Concentrations of trypsin inhibitor and immunoglobulins in colostrum of Jersey cows. J Dairy Sci 78(7) 1573-1577.
31 Telemo E, Westrom BR, Ekstrom G, Karlsson BW (1987) Intestinal macromolecular transmission in the young rat: influence of protease inhibitors during development. Biol Neonate 52(3) 141-148.
32 Filipp D, Alizadeh-Khiavi K, Richardson C, Palma A, Paredes N et al (2001) Soluble CD14 enriched in colostrum and milk induces B cell growth and differentiation. Proceed Nat Acad Sci (USA) 98(2) 603-608.
33 Murakami M, Dorschner RA, Stern LJ, Lin KH, Gallo RL (2004) Expression and secretion of cathelicidin antimicrobial peptides in murine mammary glands and human. Milk Pediatr Res 57(1) 10-15.
34 Schlimme E, Martin D, Meisel H (2000) Nucleosides and nucleotides: natural bioactive substances in milk and colostrum. Brit J Nutr 84(Suppl 1) S59-S68.
35 Carver JD, Cox WI, Barness LA (1990) Dietary nucleotide effects upon murine natural killer cell activity and macrophage activation. J Parenteral Enteral Nutr 14(1) 18-22.
36 Carver JD (1999) Dietary nucleotides: effects on the immune and gastrointestinal systems. Acta Paediatr Suppl 88(430) 83-88.
37 Donovan SM, Odle J (1994) Growth factors in milk as mediators of infant development. Ann Rev Nutr 14 147-167.
38 Borody TJ, Surace R, Warren E (2001) Tunnel vision in the bowel. Centre for Digestive Diseases, Australia.
39 Canalis E, McCarthy TL, Centrella M (1991) Growth factors and cytokines in bone cell metabolism. Annual Review of Medicine 42:17-24.
40 Canalis E, McCarthy TL, Centrella M (1989) The role of growth factors in skeletal remodeling. Endocrinol Metab Clinics North America 18(4) 903-918.
41 Molloy T, Wang Y, Murrell G (2003) The roles of growth factors in tendon and ligament healing. Sports Med 33(5) 381-394.
42 Torre (2006) op. cit.
43 Blumenfeld I, Srouji S, Lanir Y, Laufer D, Livne E (2002) Enhancement of bone defect healing in old rats by TGF-beta and IGF-1. Exp Gerontol 37(4) 553-565.
44 Adams GR, Haddad F (1996) The relationships among IGF-1, DNA content, and protein accumulation during skeletal muscle hypertrophy. J Appl Physiol 81(6) 2509-2516.
45 Rabinovsky ED (2004) The multifunctional role of IGF-1 in peripheral nerve regeneration. Neurologic Res 26(2) 204-210.
46 Xu RJ (1996) Development of the newborn GI tract and its relation to colostrum/milk intake: a review. Reproduct Fertil Developm 8(1) 35-48.
47 O'Dell SD, Day IN (1998) Insulin-like growth factor II (IGF-II). Int J Biochem Cell Biol 30(7) 767-771.
48 Bayes-Genis A, Conover CA, Schwartz RS (2000) The insulin-like growth factor axis: A review of atherosclerosis and restenosis. Circ Res 86(2) 125-130.
49 Okada M, Ohmura E, Kamiya Y, Murakami H, Onoda N et al (1991) Transforming growth factor (TGF)-alpha in human milk. Life Sci 48(12) 1151-1156.
50 Wagner CL, Forsythe DW, Pittard WB (1995) Variation in the biochemical forms of transforming growth factor-alpha present in human milk and secreted by human milk macrophages. Biol Neonate 68(5) 325-333.
51 Fallon J, Reid S, Kinyamu R, Opole I, Opole R et al (2000) In vivo induction of massive proliferation, directed migration, and differentiation of neural cells in the adult mammalian brain. Proceed Nat Acad Sci (USA) 97(26) 14686-14691.
52 Purup S, Vestergaard M, Pedersen O, Sejrsen K (2007) Biological activity of bovine milk on proliferation of human intestinal cells. J Dairy Res 74(1) 58-65.
53 Coffman RL, Lebman DA, Shrader B (1989) Transforming growth factor beta specifically enhances IgA production by lipopolysaccharide-stimulated murine B lymphocytes. J Exp Med 170(3) 1039-1044.
54 Chen SS, Li Q (1990) Transforming growth factor-beta 1 (TGF-beta 1) is a bifunctional immune regulator for mucosal IgA responses. Cellul Immunol 128(2) 353-361.
55 Centrella M, McCarthy TL, Canalis E (1988) Skeletal tissue and transforming growth factor beta. FASEB J 2(15) 3066-2073.
56 Centrella M, McCarthy TL, Canalis E (1989) Effects of transforming growth factors on bone cells. Conn Tissue Res 20(1-4) 267-275.
57 Yagi H, Suzuki S, Noji T, Nagashima K, Kuroume T (1986) Epidermal growth factor in cow's milk and milk formulas. Acta Paed Scand 75(2) 233-235.
58 Xiao X, Xiong A, Chen X, Mao X, Zhou X (2002) Epidermal growth factor concentrations in human milk, cow's milk and cow's milk-based infant formulas. Chin Med J (English) 115(3) 451-454.
59 Boonstra J, Rijken P, Humbel B, Cremers F, Verkleij A et al (1995) The epidermal growth factor. Cell Biol Int 19(5) 413-430.
60 Boonstra J, Rijken P, Humbel B, Cremers F, Verkleij A, van Bergen en Henegouwen P(1995) The epidermal growth factor. Cell Biology International 19(5):413-430.
61 Hironaka T, Ohishi H, Masaki T (1997) Identification and partial purification of a basic fibroblast growth factor-like growth factor derived from bovine colostrum. J Dairy Sci 80(3) 488-495.
62 Canalis (1989) op. cit.
63 Newburg DS (1999) Human milk glycoconjugates that inhibit pathogens. Curr Med Chem 6(2) 117-127.
64 Kawasaki Y, Isoda H, Tanimoto M, Dosako S, Idota T, Ahiko K (1992) Inhibition by lactoferrin and kappa-casein glycomacropeptide of binding of Cholera toxin to its receptor. Biosci Biotechnol Biochem 56(2) 195-198.
65 Kawasaki Y, Isoda H, Shinmoto H, Tanimoto M, Dosako S et al (1993) Inhibition by kappa-casein glycomacropeptide and lactoferrin of influenza virus hemagglutination. Biosci Biotechnol Biochem 57(7) 1214-1215.
66 Brody EP (2000) Biological activities of bovine glycomacropeptide. Brit J Nutr 84(Suppl 1) S39-S46.
67 Makowski L, Brittingham KC, Reynolds JM, Suttles J, Hotamisligil GS (2005) The fatty acid binding protein, aP2, coordinates macrophage cholesterol trafficking and inflammatory activity: Macrophage expression of aP2 impacts peroxisome proliferator-activated receptor gamma and I-kappa β kinase activities. J Biol Chem 280(13) 12888-12895.
68 Sawyer L, Kontopidis G (2000) The core lipocalin, bovine beta-lactoglobulin. Biochem Biophys Acta 1482(1-2) 136-148.
69 Superti F, Ammendolia MG, Valenti P, Seganti L (1997) Anti-rotaviral activity of milk proteins: lactoferrin prevents rotavirus infection in the enterocyte-like cell line HT-29. Med Microbiol Immunol (Berlin) 186(2-3) 83-91.
70 Ulcova-Gallova Z, Fialova P, Krauz V (1994) Immunologic factors in human colostrum and milk. Casopis Lekaru Ceskych 133(9) 275-276.
71 Sorensson J, Ohlson M, Bjornson A, Haraldsson B (2000) Orosomucoid has a cAMPdependent effect on human endothelial cells and inhibits the action of histamine. Am J Physiol - Heart Circulat Physiol 278(5) H1725-H1731.
72 Kang SW, Lim SW, Choi SH, Shin KH, Chun BG et al (2000) Antisense oligonucleotide of clusterin mRNA induces apoptotic cell death and prevents adhesion of rat ASC-17D. Sertoli cells Molecules Cells 10(2) 193-198.
73 Carver JA, Rekas A, Thorn DC, Wilson MR (2003) Small heat-shock proteins and clusterin: intra- and extracellular molecular chaperones with a common mechanism of action and function? IBUMB Health 55(12) 661-668.
74 Pons SM et al (2000) Triacylglycerol composition in colostrum, transitional and mature human milk. Eur J ClinNutr 54(12) 878-882.
75 Ronneberg R, Skara B (1992) Essential fatty acids in human colostrum. Acta Paediat 81(10) 779-783.
76 Shope RE, Gowen JW (1928) Cholesterol and cholesterol ester content of bovine. Colostrum J Exp Med 48(1) 21-24.
77 Haug A, Hostmark AT, Harstad OM (2007) Bovine milk in human nutrition - a review. Lipids Health Dis 6 25.



